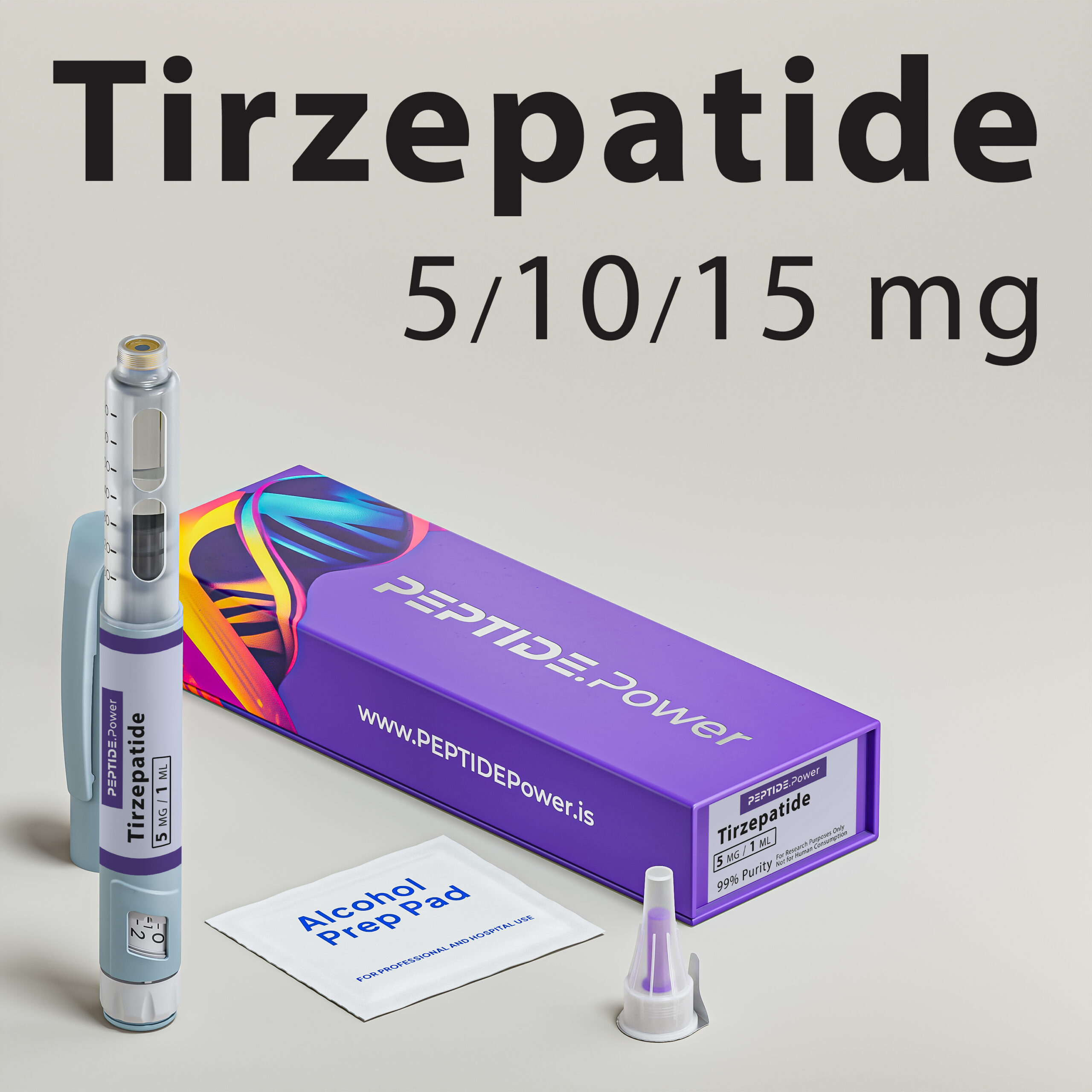

Tirzepatide
90,00 € – 235,00 €Price range: 90,00 € through 235,00 €
Tirzepatide - Dual GLP-1/GIP Agonist for Weight Loss & Metabolic Support
Description
Tirzepatide is a synthetic analog of the natural hormones GLP-1 and GIP, designed for research use in metabolic and obesity studies. Originating from pharmaceutical development, this peptide targets metabolic regulation, glucose balance, and fat metabolism.
In research models, Tirzepatide has been observed to enhance insulin secretion and reduce appetite, supporting energy balance and metabolic efficiency.
Formulated in a stabilized pre-mixed injection pen for subcutaneous use, it provides high bioavailability and ease of dosing in experimental settings.This peptide is intended for research only and not for human use. Clinical studies have shown notable effects on weight and glucose control, making it one of the most promising peptides in metabolic research.
Clinical Status:
FDA-approved for type 2 diabetes and chronic weight management (brand formulations); other uses remain research-only.
Evidence Type:
Human RCT ✔ | Observational ✔ | Animal ▣ | In vitro ▣ | Regulatory ✔
Mechanism of Action
Tirzepatide binds to GLP-1R and GIPR on pancreatic β-cells, enhancing glucose-dependent insulin release and inhibiting α-cell glucagon secretion. The synergistic dual-agonist action improves hepatic glucose output, lipid oxidation, and mitochondrial efficiency. Additionally, central appetite regulation via hypothalamic POMC/CART activation results in reduced caloric intake.
Benefits
- Dual Agonist Mechanism for Metabolic Optimization:
Tirzepatide is studied as a dual agonist of the GLP-1 and GIP receptors, providing simultaneous stimulation of two key incretin pathways that regulate insulin secretion, appetite, and energy balance. This dual mechanism enables superior glucose regulation and energy expenditure compared to single agonists, making it one of the most advanced compounds in metabolic peptide research. - Significant Weight Reduction in Research Models:
In human and animal studies, Tirzepatide has demonstrated marked body weight reduction through decreased caloric intake and enhanced lipid metabolism. Reported findings indicate up to 22% average weight loss in long-term trials, associated with improved metabolic flexibility and reduced visceral fat accumulation. These effects are of high interest for obesity and body composition research. - Improved Glucose Control and Insulin Sensitivity:
Tirzepatide enhances pancreatic insulin secretion and suppresses glucagon in a glucose-dependent manner. It has been observed to lower fasting glucose and HbA1c levels significantly in both diabetic and non-diabetic research participants. These improvements stem from coordinated incretin receptor activation that improves beta-cell function and peripheral insulin sensitivity. - Reduction of Visceral and Hepatic Fat:
Preclinical and clinical data demonstrate that Tirzepatide reduces visceral adipose tissue and liver fat content by modulating lipid oxidation and suppressing de novo lipogenesis. This mechanism contributes to reduced hepatic steatosis and improved metabolic liver function, positioning it as a leading peptide in experimental models of fatty liver and metabolic syndrome. - Enhanced Lipid Profile and Cardiometabolic Benefits:
Research outcomes indicate a significant reduction in total cholesterol, LDL, and triglycerides with concurrent elevation of HDL levels. Tirzepatide’s effect on lipid metabolism and vascular function supports its potential use in studies focused on cardiovascular health and long-term metabolic optimization. - Regulation of Appetite and Satiety Hormones:
By modulating hypothalamic pathways and gut hormone signaling, Tirzepatide influences appetite control and satiety perception. This leads to lower caloric intake without inducing compensatory hunger or muscle catabolism. Such behavioral and neuroendocrine modulation is a critical area of ongoing research in weight regulation and eating behavior studies. - Improved Energy Expenditure and Mitochondrial Efficiency:
Studies indicate that Tirzepatide increases energy expenditure by activating thermogenic processes in brown adipose tissue and enhancing mitochondrial efficiency. These outcomes reflect a systemic improvement in metabolic energy flow, supporting its inclusion in research on metabolic performance and energy utilization optimization. - Reduction of Inflammatory Markers:
In metabolic research models, Tirzepatide administration has been associated with decreased levels of inflammatory cytokines such as CRP, TNF-α, and IL-6. This anti-inflammatory effect is attributed to improved insulin sensitivity and lipid metabolism, which collectively reduce metabolic stress and systemic inflammation. - Preservation of Lean Muscle During Weight Loss:
Unlike many calorie-restriction interventions, Tirzepatide supports the maintenance of lean muscle mass during fat reduction. This effect is vital in research evaluating sustainable body recomposition and metabolic health, as it preserves basal metabolic rate and muscular strength while decreasing total adiposity. - Synergistic Effects with Other Metabolic Peptides:
Experimental designs often explore the combination of Tirzepatide with Cagrilintide, Semaglutide, or AOD-9604 to evaluate enhanced effects on lipid metabolism, appetite regulation, and glucose balance. These synergistic models aim to amplify fat loss outcomes and expand understanding of dual and triple incretin receptor activation in metabolic research. - Long-Term Metabolic Adaptation:
Extended-duration studies reveal that Tirzepatide maintains metabolic improvements beyond the treatment period, suggesting sustained changes in energy homeostasis. Its influence on gene expression related to adipogenesis, mitochondrial activity, and glucose transport underscores its long-term potential in metabolic regulation research.
Research Data
| Study / Model | Reported effect |
| Human clinical trials (SURPASS program, T2DM) |
↓ HbA1c (up to -2.4%) and body weight (-12 kg over 40 weeks) vs insulin and Semaglutide.
|
| Human clinical trials (obesity without diabetes) |
15-22% mean weight loss at 72 weeks; improved lipids and blood pressure.
|
| Comparative RCTs (Tirzepatide vs Semaglutide) |
Higher efficacy in weight and glycemic outcomes; similar tolerability.
|
| NAFLD / NASH models |
↓ hepatic steatosis and transaminases (ALT/AST); improved insulin response and mitochondrial function.
|
| GLP-1/GIP receptor studies (preclinical) |
Dual receptor activation → ↑ insulin secretion, ↓ appetite, ↑ energy efficiency.
|
| Cardiometabolic outcomes (post-hoc human) |
↓ total cholesterol, LDL, triglycerides, CRP; improved insulin sensitivity.
|
| Animal obesity / high-fat diet models |
↓ fat mass, ↑ thermogenesis and glucose tolerance.
|
| Human tolerability studies (Phases 1-3) |
Common mild effects: nausea, diarrhea; no serious hepatic or hematologic effects.
|
| Long-term weight maintenance (extension trials) |
>80% of weight reduction maintained after 88 weeks of continued use.
|
Stack Suggestions
Tirzepatide is often stacked with:
- Cagrilintide → synergistic appetite suppression and fat loss via complementary amylin pathway
- 5-Amino-1MQ → enhanced mitochondrial thermogenesis and energy expenditure.
GHK-Cu + BPC-157 stack → supports tissue recovery and insulin sensitivity improvement during weight loss - NAD+ or MOTS-c → improves mitochondrial efficiency and energy balance.
Stacks discussed are for experimental design; not safety/efficacy guidance.
Pen Dosage Chart
| Tirzepatide Pen 5 mg | |
| Volume | 1.0 mL |
| mg/mL | 5.0 mg/mL |
| Click-to-Dose | 1 click = 0.05 mg |
| Example(s) | 10 clicks = 0.5 mg; 50 clicks = 2.5 mg |
| Tirzepatide Pen 10 mg | |
| Volume | 1.0 mL |
| mg/mL | 10.0 mg/mL |
| Click-to-Dose | 1 click = 0.10 mg |
| Example(s) | 10 clicks = 1 mg; 25 clicks = 2.5 mg |
| Tirzepatide Pen 15 mg | |
| Volume | 1.5 mL |
| mg/mL | 10.0 mg/mL |
| Click-to-Dose | 1 click = 0.10 mg |
| Example(s) | 10 clicks = 1 mg; 25 clicks = 2.5 mg |
Dosage & Protocols Variations
Standard Research Protocol
- Dose: 2.5 – 5 mg
- Duration: 8 – 12 weeks
- Frequency: 1× weekly
- Cycle Interval: Optional 2-week pause
- Goal / Description: For basic metabolic modulation studies
Therapeutic Research Protocol
- Dose: 10 – 15 mg
- Duration: 24 – 48 weeks
- Frequency: 1× weekly
- Cycle Interval: Repeat after 1 month
- Goal / Description: Modeled on SURPASS/SURMOUNT designs; for long-term metabolic outcomes
Biohacker Microdosing
- Dose: 0.5 – 1 mg every 3 days
- Duration: 4 – 8 weeks
- Frequency: Every 3 days
- Cycle Interval: Repeat after 1 month
- Goal / Description: Used experimentally to assess mild appetite control and metabolic priming
Maintenance Phase
- Dose: 2.5 – 5
- Duration: Variable
- Frequency: 1× weekly
- Cycle Interval: Continuous
- Goal / Description: Sustain achieved body composition and metabolic balance.
Possible Side Effects
Tirzepatide may cause mild and transient gastrointestinal symptoms in some research subjects.
The most common reactions include nausea, diarrhea, and reduced appetite, particularly during initial exposure. Occasional reports include constipation, vomiting, or abdominal discomfort, typically resolving as tolerance develops. Other possible effects include fatigue or transient increases in heart rate during activity.
All effects are reversible and dose-dependent. Proper titration minimizes occurrence.
Product Attributes
- CAS #: 2023788-19-2
- Molecular Formula: C225H348N48O68
- Sequence (AA): Modified 39-amino-acid linear peptide (GLP-1/GIP analog)
- Molecular Weight: ~4813.6 g/mol
- PubChem CID: 155090095
- Half-Life: ~120 hours (≈5 days)
- Synonyms: LY3298176, GLP-1/GIP Dual Agonist, GLP-1T, GIP/GLP-1 RA, Twincretin
- Type: Synthetic peptide analog
- Research Focus: Weight Loss, Metabolic Health, Cardiometabolic Optimization
Scientific References
- Tirzepatide once weekly for the treatment of obesity Human RCT
- Tirzepatide: a systematic update Review
- Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes Human RCT
- Efficacy and safety of once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin in patients with type 2 diabetes (SURPASS-3): a randomised, open-label, parallel-group, phase 3 trial Human RCT
- Dual GIP and GLP-1 receptor agonist tirzepatide improves beta-cell function and insulin sensitivity in type 2 diabetes Human observational
- Effects of novel dual GIP and GLP-1 receptor agonist tirzepatide on biomarkers of inflammation and cardiovascular risk in patients with type 2 diabetes Human RCT
- LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: from discovery to clinical proof of concept Animal
- A long-term safety study of tirzepatide (LY3298176) in participants with type 2 diabetes Human RCT
- Effect of tirzepatide versus insulin degludec on liver fat content and abdominal adipose tissue in people with type 2 diabetes (SURPASS-3 MRI): a substudy of the randomised, open-label, parallel-group, phase 3 SURPASS-3 trial Human RCT
- Once-weekly semaglutide in adolescents with obesity (comparative to tirzepatide context) Human RCT